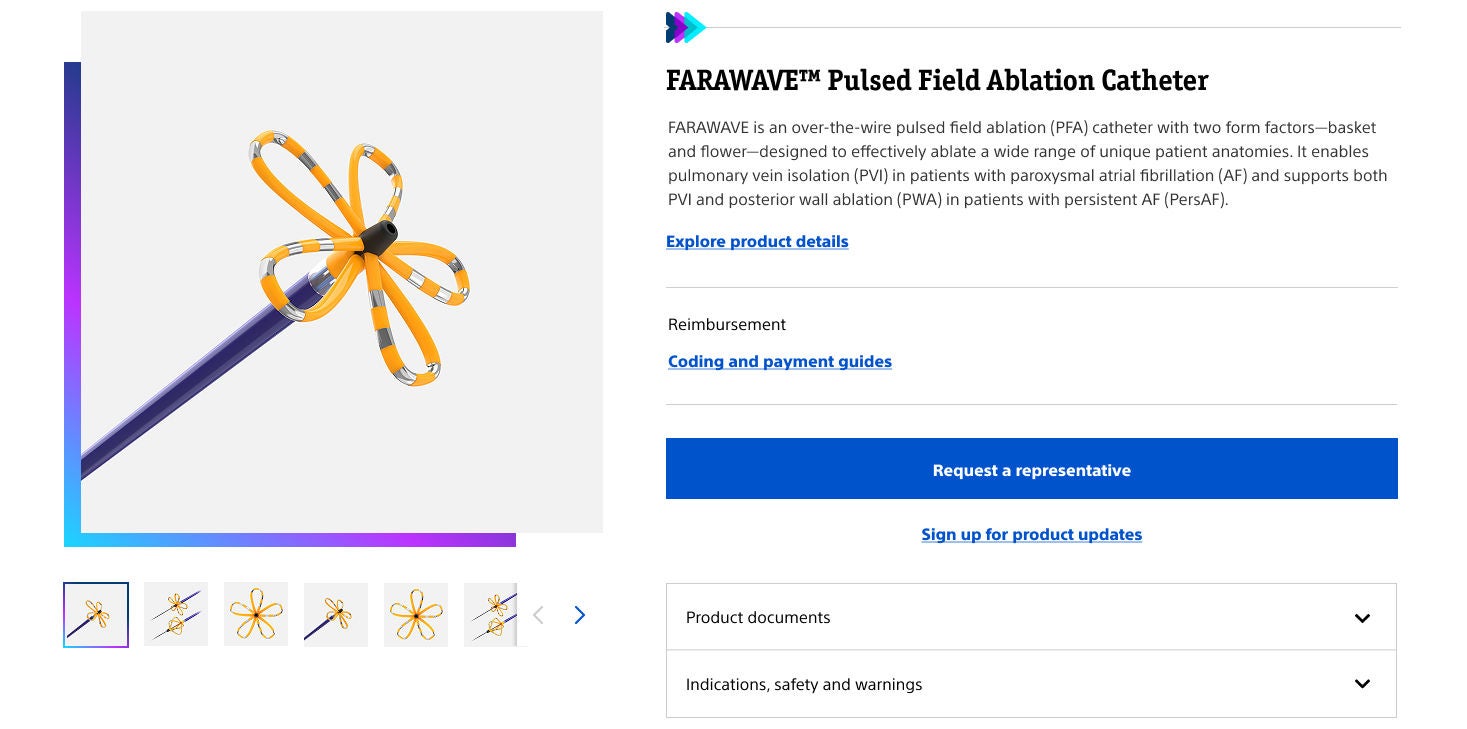
- Overview
- Clinical data
- Technical specifications
- Ordering information
- Training
- Resources
Benefit-driven marketing message for this product
Very brief description - ut enim ad minim veniam, quis nostrud exercitation ullamco laboris nisi ut aliquip ex ea commodo consequat.

How the product works
Describe the video showing how the system works lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Ut enim ad minim veniam, quis nostrud exercitation ullamco laboris nisi ut aliquip ex ea commodo consequat.
Why choose this product
The most advanced system yet

Key differentiators
5 min
procedures that use this product are performed every day.
78%
of patients saw improvements in the first 6 weeks.
~30K
procedures that use this product are performed every day.
What patient populations are a good fit for this product?
This product sees positive outcomes in patients with isolation of pulmonary veins and the posterior wall in the treatment of drug-refractory, symptomatic Persistent Atrial Fibrillation (episode duration less than one year).
This product sees positive outcomes in patients with isolation of pulmonary veins and the posterior wall in the treatment of drug-refractory, symptomatic Persistent Atrial Fibrillation (episode duration less than one year).
This product sees positive outcomes in patients with isolation of pulmonary veins and the posterior wall in the treatment of drug-refractory, symptomatic Persistent Atrial Fibrillation (episode duration less than one year).

What's included with this product?
- VersaCross RF Wire (j-tip or pigtail)
- Single-use RFP-100A™ Connector Cable
- VersaCross Transseptal Sheath (8.5 F)
- Dilator with TRUform™ shapeable technology
Callout for a new or featured clinical data study or trial
Clinical overview / messaging
Add some copy that summarizes or highlights clinical benefits of this product that also supports SEO. Ut enim ad minim veniam, quis nostrud exercitation ullamco laboris nisi ut aliquip ex ea commodo consequat.
74%
impactful stat demonstrating a clinical benefit
300K
impactful stat demonstrating a clinical benefit
~50m
impactful stat demonstrating a clinical benefit
Explore all clinical data
Real-world data
Example Clinical Trial Name of Title of the Study
Reference- REDDY et. al., 2024
Add some copy that introduces users to the flagship product. This could be benefit driven messaging or something more technical. This is mainly needed to support the layout of the page, so add something with solid SEO value if you can. Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Ut enim ad minim veniam, quis nostrud exercitation ullamco laboris nisi ut aliquip ex ea commodo consequat.
74%
impactful stat demonstrating a clinical benefit
300K
impactful stat demonstrating a clinical benefit
~50m
impactful stat demonstrating a clinical benefit
Real-world data
Example Clinical Trial Name of Title of the Study
Reference- REDDY et. al., 2024
Add some copy that introduces users to the flagship product. This could be benefit driven messaging or something more technical. This is mainly needed to support the layout of the page, so add something with solid SEO value if you can. Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Ut enim ad minim veniam, quis nostrud exercitation ullamco laboris nisi ut aliquip ex ea commodo consequat.
74%
impactful stat demonstrating a clinical benefit
300K
impactful stat demonstrating a clinical benefit
~50m
impactful stat demonstrating a clinical benefit
Real-world data
Example Clinical Trial Name of Title of the Study
Reference- REDDY et. al., 2024
Add some copy that introduces users to the flagship product. This could be benefit driven messaging or something more technical. This is mainly needed to support the layout of the page, so add something with solid SEO value if you can. Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Ut enim ad minim veniam, quis nostrud exercitation ullamco laboris nisi ut aliquip ex ea commodo consequat.
74%
impactful stat demonstrating a clinical benefit
300K
impactful stat demonstrating a clinical benefit
~50m
impactful stat demonstrating a clinical benefit
Real-world data
Example Clinical Trial Name of Title of the Study
Reference- REDDY et. al., 2024
Add some copy that introduces users to the flagship product. This could be benefit driven messaging or something more technical. This is mainly needed to support the layout of the page, so add something with solid SEO value if you can. Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Ut enim ad minim veniam, quis nostrud exercitation ullamco laboris nisi ut aliquip ex ea commodo consequat.
74%
impactful stat demonstrating a clinical benefit
300K
impactful stat demonstrating a clinical benefit
~50m
impactful stat demonstrating a clinical benefit
Technical specifications
| Feature | Specifications |
| Tip configurations | J-tip, pigtail |
| Curve shape | 9 mm (j-tip), 24 mm (pigtail) |
| Wire diameter | 0.035 in (0.89 mm) |
| Overall length | 180 cm, 230 cm |
Ordering information
| UPN | GTIN | Size (Fr x cm) | Material Type | Suture Type |
|---|---|---|---|---|
| M0061902050 | 08714729937715 | 4.8 X 10 | Firm | Braided |
| M0061902060 | 08714729937722 | 4.8 x 12 | Firm | Braided |
| M0061902070 | 08714729937739 | 4.8 x 14 | Firm | Braided |
| M0061902080 | 08714729937746 | 4.8 x 16 | Firm | Braided |
| M0061902090 | 08714729937753 | 4.8 x 18 | Firm | Braided |
| M0061902100 | 08714729937760 | 4.8 x 20 | Firm | Braided |
| M0061902110 | 08714729937777 | 4.8 x 22 | Firm | Braided |
| M0061902120 | 08714729937784 | 4.8 x 24 | Firm | Braided |
| M0061902130 | 08714729937791 | 4.8 x 26 | Firm | Braided |
| M0061902140 | 08714729937807 | 4.8 x 28 | Firm | Braided |
| M0061902150 | 08714729937814 | 4.8 x 30 | Firm | Braided |
| M0061902200 | 08714729937821 | 6 x 20 | Firm | Monofilament |
| M0061902210 | 08714729937838 | 6 x 22 | Firm | Monofilament |
How to order this product
Boston Scientific E-Commerce
Add some copy that descirbes the purchase process for e-commerce.
Ordering through customer support
Add some copy that describes the purchase process for customer support.
Training overview / messaging
Add some copy for SEO: Lorem ipsum dolor sit amet, consectetur adipiscing elit, sed do eiusmod tempor incididunt ut labore et dolore magna aliqua. Ut enim ad minim veniam, quis nostrud exercitation ullamco laboris nisi ut aliquip ex ea commodo consequat.
Featured training

Duration: 1 hour
Course description: provides information about the LUX-Dx II/II+ ICMs* and LATITUDE Clarity™ Data Management System. It focuses on LUX-Dx II and LUX-Dx II+.

Duration: 1 hour
Course description: provides information about the LUX-Dx II/II+ ICMs* and LATITUDE Clarity™ Data Management System. It focuses on LUX-Dx II and LUX-Dx II+.

Duration: 1 hour
Course description: provides information about the LUX-Dx II/II+ ICMs* and LATITUDE Clarity™ Data Management System. It focuses on LUX-Dx II and LUX-Dx II+.

Duration: 1 hour
Course description: provides information about the LUX-Dx II/II+ ICMs* and LATITUDE Clarity™ Data Management System. It focuses on LUX-Dx II and LUX-Dx II+.
Online medical training and education courses
The EDUCARE online platform makes healthcare education and training more relevant, more comprehensive, more personal, and more accessible. Register to access a library of procedural videos, case studies, training resources, and events.
Callout for a new or featured resource
Sign up for product updates
Get more information about this product and find out how you can start using it to treat your patients.
Frequently asked questions
This is some placeholder text that answers the question. The answer is descriptive to better support SEO and GEO.
This is some placeholder text that answers the question. The answer is descriptive to better support SEO and GEO.
This is some placeholder text that answers the question. The answer is descriptive to better support SEO and GEO.
This is some placeholder text that answers the question. The answer is descriptive to better support SEO and GEO.
This is some placeholder text that answers the question. The answer is descriptive to better support SEO and GEO.